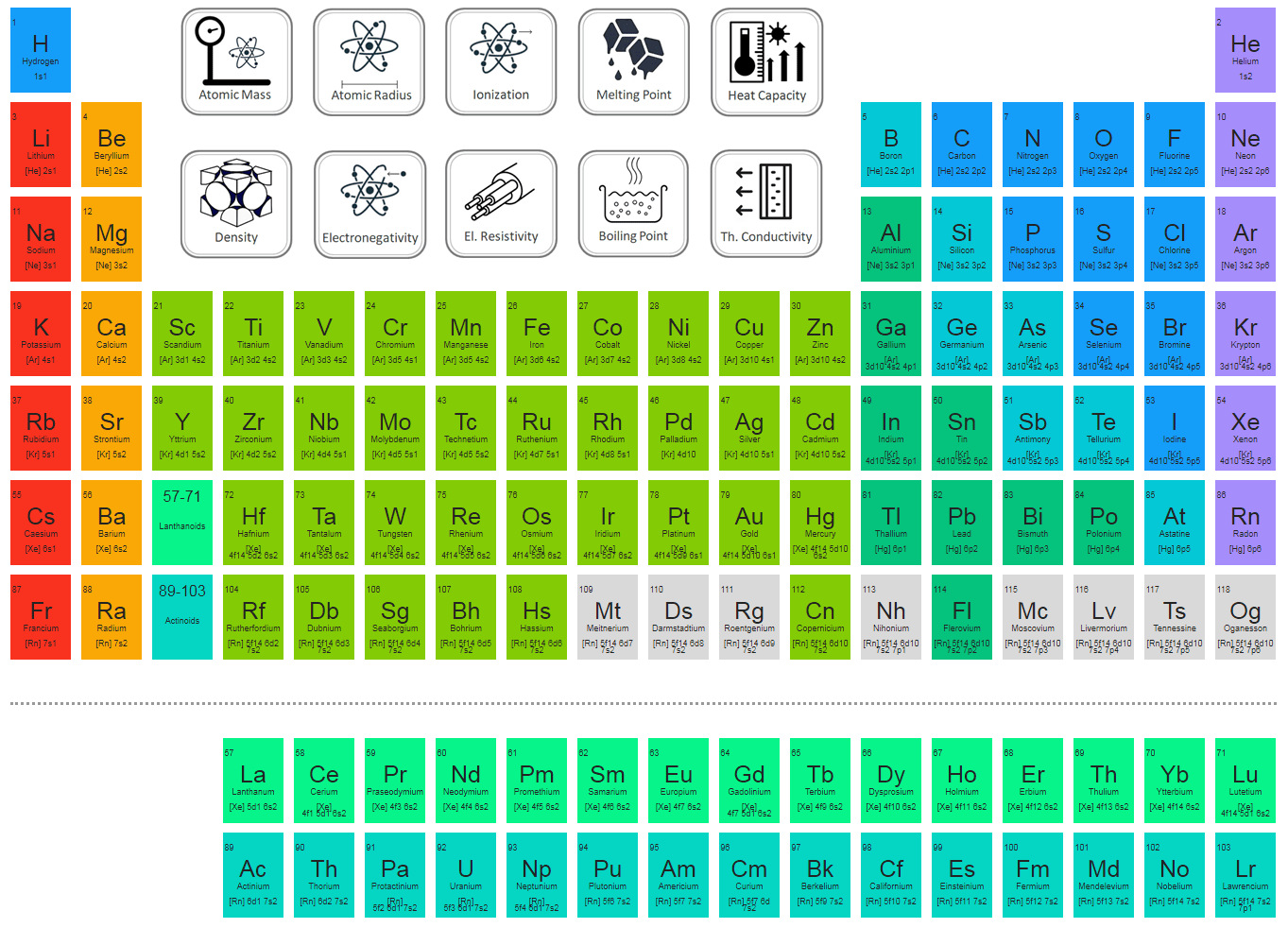
Group 1 (the alkali metals) is an example. Preserve the principle of classification by keeping elements with similarĮlements in the same vertical column or group have similar electronicĬonfigurations, have the same number of electrons in the outer orbitals, and Placed separately in the periodic table to maintain its structure and to The 4f- and 5f- transition series of elements are Gives the 5f-inner transition series known as the Actinoid Series. Filling up of the 5f orbitals after actinium (Z=89) This period will end at the element with atomic number 118 which would belong (n=7) is similar to the sixth period with the successive filling up of the 7s,ĥf, 6d and 7p orbitals and includes most of the man-made radioactive elements. Lutetium (Z=71) to give the 4f-inner transition series, which is called the Lanthanoid Series. Filling up of the 4f orbitals begins with cerium (Z=58) and ends at The sixth period (n=6) contains 32Įlements and successive electrons enter 6s, 4f, 5d and 6p orbitals, in that This period endsĪt xenon with filling up of the 5p orbitals. Period (n=5) beginning with rubidium is similar to the fourth period andĬontains the 4d transition series starting at yttrium (Z=39). Altogether we have 18 elements in this fourth period. The fourth period ends at krypton with the filling up of the 4p Note that before the 4p orbital is filled, filling up of 3d orbitals becomesĮnergetically favourable and we come across the so-called 3d Transition Series Period (n=4) starts at potassium with the filling up of 4s orbital. Gives rise to the third period of 8 elements from sodium to argon. The third period (n=3) begins at sodium, and theĪdded electron enters a 3s orbital. 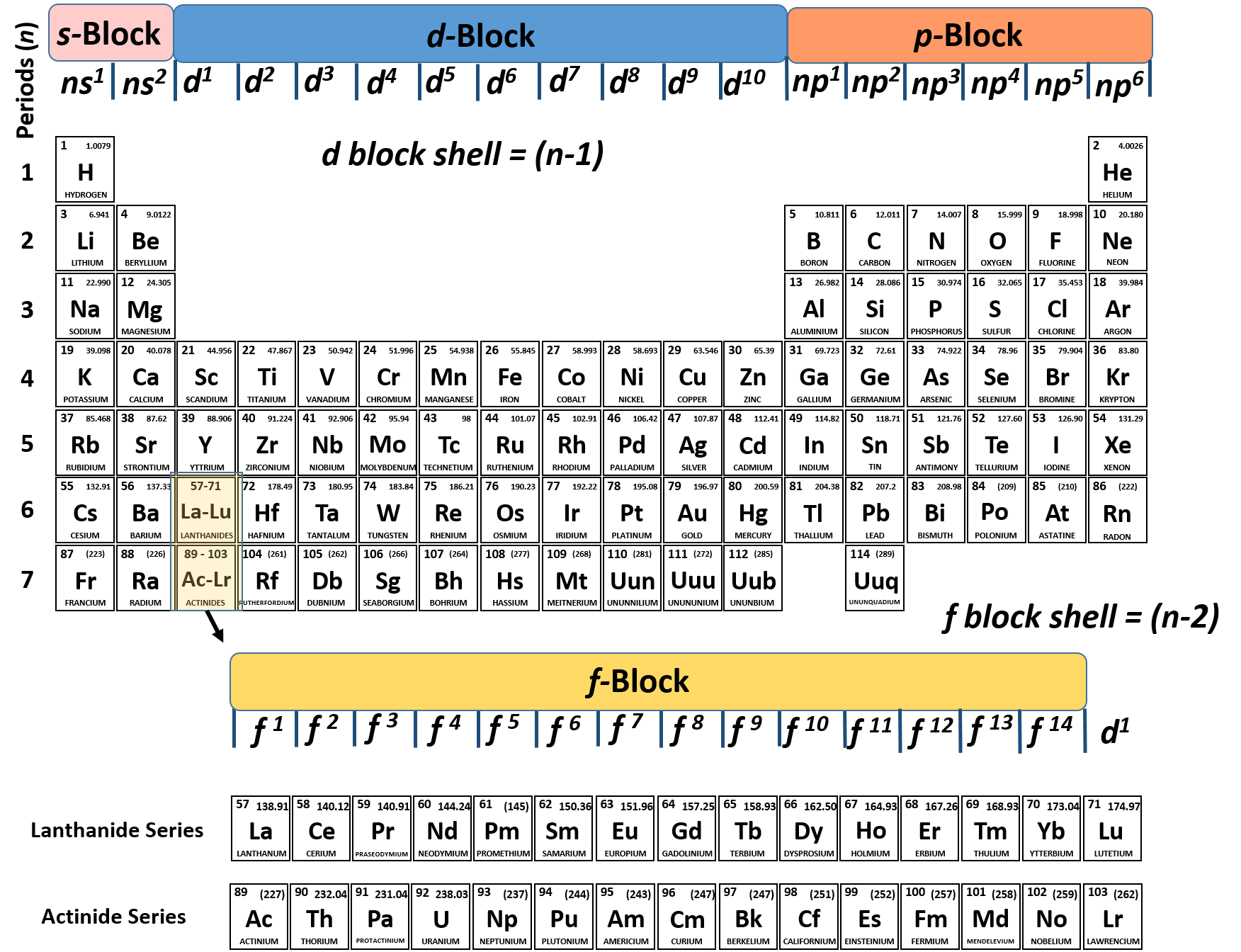
Thus there are 8Įlements in the second period. Startingįrom the next element boron, the 2p orbitals are filled with electrons when the The next element, beryllium has four electronsĪnd has the electronic configuration 1s 22s 2. The second period starts with lithium and the thirdĮlectron enters the 2s orbital. Period starts with the filling of the lowest level (1s) and has thus the twoĮlements-hydrogen (1s 1) and helium (1s 2) when the first Readily seen that the number of elements in each period is twice the number ofĪtomic orbitals available in the energy level that is being filled. The electronic configuration ofĮlements can be best studied in terms of variations in periods and groups ofĮach successive period in the periodic table is associated with theįilling up of the next higher principal energy level (n=1, n=2,etc.). The principal quantum number (n) defines the main energy level known as the Shell. We have already learnt thatĪn electron in an atom is characterized by a set of four quantum numbers and There is a close connection between the electronic configuration of theĮlements and the long form of the Periodic Table. Electronic configuration and periodic table1
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
